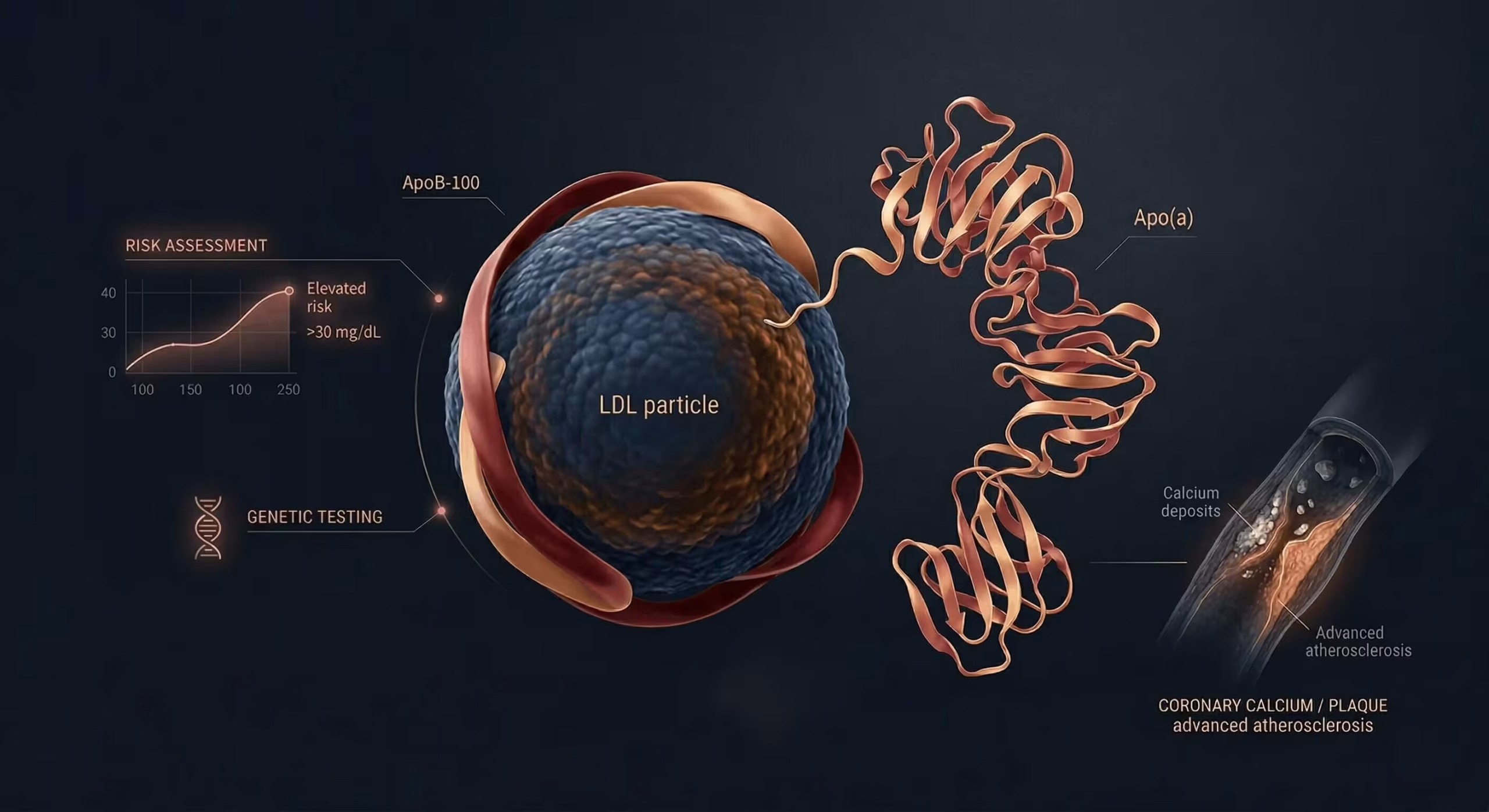
Lipoprotein(a), or Lp(a), is a genetically determined LDL-like particle that increases the risk of atherosclerotic cardiovascular disease and calcific aortic valve stenosis. Because Lp(a) is not included in a standard lipid panel and is only minimally affected by lifestyle, it can remain hidden unless specifically measured. Despite affecting roughly one in five people worldwide, Lp(a) remains undertested in routine practice. Current guidance recommends measuring Lp(a) at least once in adulthood, because an elevated level can change how aggressively LDL-C and other modifiable cardiovascular risk factors should be managed.
- Measure Lp(a) at least once in adulthood
- Consider ≥125 nmol/L (≥50 mg/dL) a high-risk threshold
- Use Lp(a) as a risk-enhancing factor, not a routine treatment target
- Pair Lp(a) with PREVENT, CAC, LDL-C, and sometimes ApoB for better risk assessment
- Phase 3 outcome trials for pelacarsen, olpasiran and lepodisiran are expected to report from 2026
For a plain-English summary, see our patient guide to high Lp(a). For the broader guideline framework, see our 2026 dyslipidemia guideline summary.
Why Lp(a) matters clinically
Lp(a) captures inherited cardiovascular risk that is often missed by the standard lipid panel. High Lp(a) is associated with coronary artery disease, ischaemic stroke, peripheral artery disease, and calcific aortic valve stenosis. Importantly, patients with apparently well-controlled LDL-C may still carry substantial residual risk if Lp(a) is markedly elevated. On a per-particle basis, an Lp(a) particle is estimated to be approximately six-fold more atherogenic than an LDL particle (Björnson et al., JACC 2024).
This is one of the reasons Lp(a) has moved from a niche specialist test to a mainstream part of cardiovascular risk assessment. For many patients, the value of the result is not that it changes one lab number, but that it changes how seriously overall risk is taken and how intensive preventive therapy becomes. The 2026 ACC/AHA dyslipidemia guideline now explicitly includes Lp(a) as a risk-enhancing factor that can influence treatment decisions.
Who should be tested for Lp(a)?
Routine testing
Current cardiovascular guidance — including from the National Lipid Association and the 2026 ACC/AHA guideline — recommends measuring Lp(a) at least once in adulthood. Because levels are largely genetically determined and usually stable over time, a single measurement is enough for most people (Class of Recommendation I, Level of Evidence B-NR).
When testing is especially helpful
Lp(a) testing is particularly useful in people with premature cardiovascular disease (before age 55 in men, 65 in women), a strong family history of early heart disease, known or suspected familial hypercholesterolaemia, unexplained residual risk despite well-controlled LDL-C, borderline or intermediate overall risk where treatment decisions are uncertain, or calcific aortic valve disease. In children, selective screening is recommended when there is a family history of FH, premature ASCVD, or a first-degree relative with elevated Lp(a). Our FH calculator can help structure the assessment for inherited lipid disorders.
How should Lp(a) be measured and interpreted?
Preferred units
Lp(a) is best interpreted in the units reported by the laboratory. When available, nmol/L is generally preferred because it reflects particle number more directly and is less distorted by apolipoprotein(a) size variability than mg/dL. Immunochemical assays calibrated against the WHO/IFCCLM reference material are the recommended method.
Why mg/dL and nmol/L cannot be reliably converted
Unlike standard cholesterol measures, Lp(a) does not have a dependable fixed conversion factor between mg/dL and nmol/L. The apo(a) component varies substantially in size due to kringle IV type 2 (KIV-2) repeat polymorphism — from 3 to over 40 copies — so the same mass concentration may represent different particle numbers. For this reason, results should be interpreted in the units provided by the assay.
Practical interpretation of levels
(<30 mg/dL)
(30–50 mg/dL)
(≥50 mg/dL)
(~100 mg/dL)
Risk is continuous, not all-or-none. A value just below a threshold is not "normal" in an absolute sense, and a very high value matters more in someone who also has elevated LDL-C, diabetes, hypertension, smoking, or coronary calcium. Levels around 250 nmol/L are associated with roughly double the long-term risk of cardiovascular events. This is where the next section — integrating Lp(a) with other risk tools — becomes particularly useful.
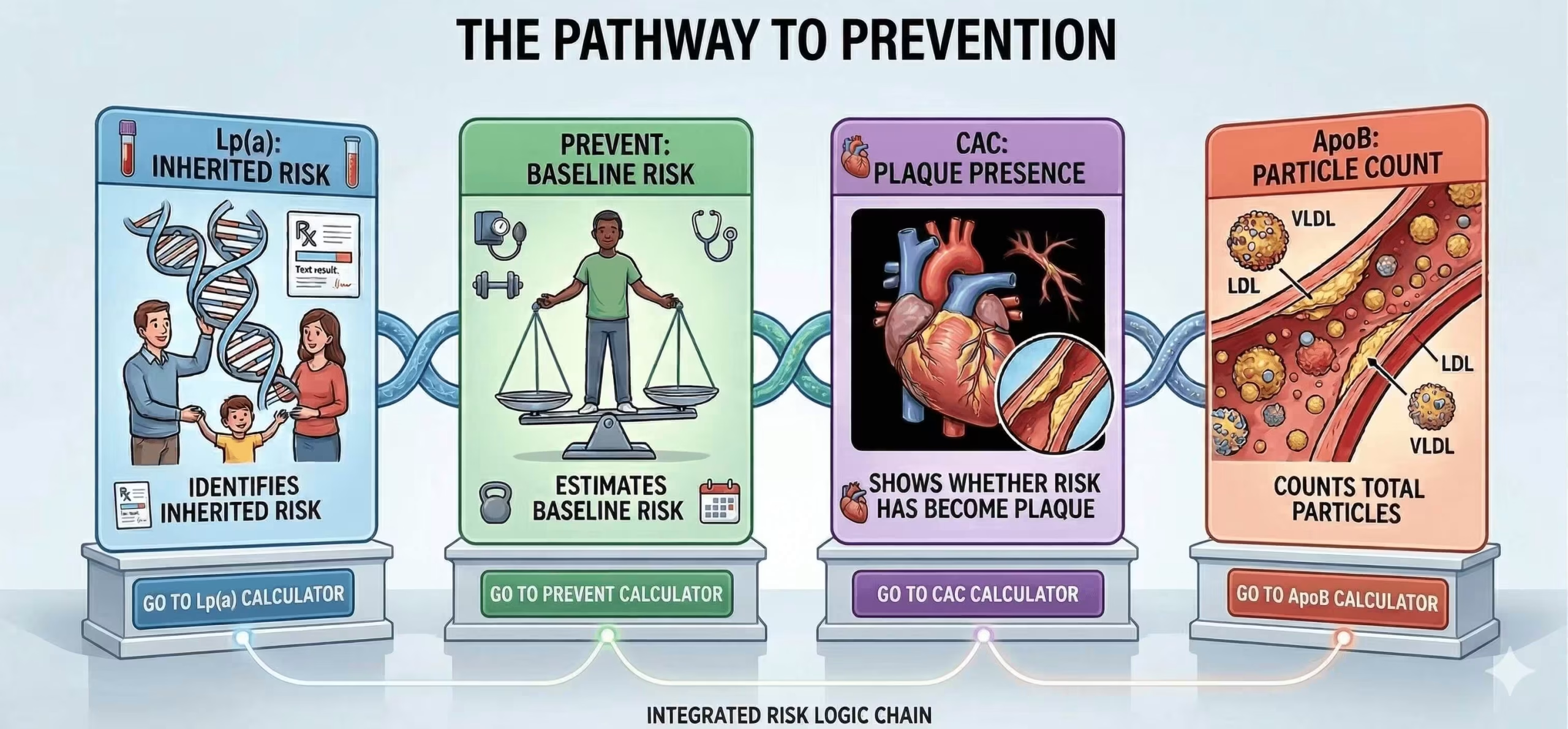
How elevated Lp(a) changes risk assessment: PREVENT, CAC and ApoB
An elevated Lp(a) result should not be viewed in isolation. Its real value is in changing the interpretation of overall cardiovascular risk — especially in patients whose risk estimate is borderline, whose LDL-C appears reasonable, or whose family history suggests more risk than the standard lipid panel shows.
PREVENT
The PREVENT equations are now the preferred tool for primary prevention risk estimation in adults aged 30–79 years. Lp(a) is not a replacement for PREVENT; rather, it adds context to the estimated risk and may justify a lower threshold for intensifying prevention when inherited risk is clearly elevated.
Coronary artery calcium (CAC)
If treatment decisions remain uncertain after standard risk assessment, coronary artery calcium scoring can help reclassify risk. In the 2026 dyslipidemia guideline, selective CAC use is recommended in borderline or intermediate-risk patients, and the result can influence LDL-C goals and treatment intensity. In practice, Lp(a) and CAC work well together: Lp(a) identifies inherited risk, while CAC shows whether that risk has already translated into measurable plaque burden. For more on how calcium scoring is used clinically, see our dedicated article.
ApoB
Lp(a) and ApoB are related, but they are not interchangeable. Each Lp(a) particle carries one ApoB-100 molecule, but ApoB reflects the total number of atherogenic particles across LDL, IDL, VLDL remnants, and Lp(a). That makes ApoB especially useful when LDL-C appears acceptable but residual risk remains high — for example in diabetes, metabolic syndrome, hypertriglyceridaemia, chronic kidney disease, or established cardiovascular disease. Where LDL-C and ApoB are discordant, ApoB is generally the better guide to atherogenic burden.
Management of elevated Lp(a)
A high Lp(a) result usually does not mean there is a specific Lp(a)-targeted treatment to start today. Instead, it means the patient's overall cardiovascular risk should be taken more seriously. In most cases, the practical response is to lower LDL-C more intensively, treat blood pressure and diabetes carefully, stop smoking, and consider additional risk refinement with ApoB or CAC if management remains uncertain.
LDL-C lowering: statins, ezetimibe and PCSK9 inhibitors
Statins do not directly lower Lp(a) — and may cause a modest increase — but they still reduce cardiovascular risk and remain foundational therapy when LDL-C lowering is indicated. Ezetimibe may be added when LDL-C goals are not achieved with statins alone. PCSK9 inhibitors produce a modest Lp(a) reduction of approximately 20–30% in addition to their potent LDL-C lowering, and may be considered in high-risk patients who do not reach target on maximally tolerated statin plus ezetimibe. For patients with familial hypercholesterolaemia, PCSK9-based therapy may be particularly important when both LDL-C and Lp(a) are elevated.
Lipoprotein apheresis
Apheresis remains a niche but important option in selected very high-risk patients — particularly those with FH, established ASCVD or PAD, Lp(a) ≥60 mg/dL (~150 nmol/L), and LDL-C ≥100 mg/dL despite maximally tolerated therapy. Apheresis can lower both LDL-C and Lp(a) by 60–70% acutely, with a time-averaged Lp(a) reduction of approximately 30–35%.
Repeat testing and family screening
Because Lp(a) is largely genetic and stable, repeat testing is usually unnecessary unless there is a specific clinical reason (such as changes in renal function or menopause). An elevated result should prompt consideration of cascade screening in first-degree relatives, especially when there is a family history of premature ASCVD or familial hypercholesterolaemia.
Aspirin
Aspirin should not be presented as routine Lp(a)-directed therapy. If considered, it should be framed within standard primary or secondary prevention decision-making, weighing cardiovascular benefit against individual bleeding risk.
Emerging Lp(a)-lowering therapies
Targeted Lp(a) lowering is one of the most active areas in preventive cardiology. Several RNA-based therapies have demonstrated reductions of 80–94% in phase 2 trials, and major phase 3 cardiovascular outcome trials are now underway. Definitive event-reduction data are still pending — meaning Lp(a) is already clinically important today, but routine treatment still focuses on LDL-C and overall risk reduction until outcome trials report.
Trial: Lp(a)HORIZON · Phase 3 · n=8,323
Results expected: H1 2026
Phase 2: ~80% Lp(a) reduction
Dosing: 80 mg SC monthly
Trial: OCEAN(a) · Phase 3 · n>7,200
Results expected: Late 2026–2027
Phase 2: ~94% Lp(a) reduction
Dosing: SC every 12 weeks
Trial: ACCLAIM-Lp(a) · Phase 3 · n≈12,500
Results expected: ~2029
Phase 2: 94% reduction at 180 days (single dose)
Dosing: SC once or twice yearly
Trial: KRAKEN · Phase 2 completed
Status: Phase 3 planning
Phase 2: Up to 86% Lp(a) reduction
Dosing: Oral daily
Special populations and clinical considerations
The clinical implications of elevated Lp(a) differ depending on the patient's baseline risk, age, comorbidities, and family history. In some patients it serves mainly as a risk-enhancer in primary prevention; in others — especially those with FH, established ASCVD, or calcific aortic valve disease — it may help explain why risk is higher than expected.
Primary prevention
In adults aged 40–75 years with intermediate ASCVD risk, Lp(a) levels ≥125 nmol/L (≥50 mg/dL) are considered risk-enhancing factors that may support initiation or intensification of statin therapy. The PREVENT calculator can help frame baseline risk in this group.
Secondary prevention
Elevated Lp(a) remains a significant contributor to residual cardiovascular risk in patients with established ASCVD, even those on optimal therapy. More aggressive LDL-C lowering with ezetimibe or PCSK9 inhibitors may be appropriate in these patients.
Familial hypercholesterolaemia
Up to 30–50% of patients with FH have co-elevated Lp(a), substantially compounding their cardiovascular risk. Lp(a) measurement is recommended in all FH patients to better stratify risk and guide more aggressive treatment. Our FH calculator can help formalise the diagnosis.
Children and adolescents
Selective screening is recommended in high-risk children, particularly those with FH or a strong family history of premature ASCVD. Early identification allows for long-term management strategies focused on reducing other modifiable risk factors.
Women's cardiovascular health
Lp(a) levels can rise during pregnancy and after menopause. A 2024 study from the Women's Health Study of nearly 28,000 initially healthy women with 30-year follow-up showed that elevated Lp(a) independently predicted first major cardiovascular events, reinforcing that Lp(a) adds long-term prognostic information even when measured once in mid-life. Hormone replacement therapy is not recommended for lowering Lp(a) due to other cardiovascular risks.
Chronic kidney disease
Lp(a) levels tend to increase as kidney function declines and contribute to the elevated cardiovascular risk in CKD. Dialysis does not effectively lower Lp(a). This may warrant periodic monitoring in patients with changing renal function.
Calcific aortic valve disease
Elevated Lp(a) is associated with calcific aortic valve stenosis through oxidised phospholipid biology and microthrombi deposition. The ongoing Lp(a)FRONTIERS CAVS trial is testing whether Lp(a) lowering can slow valve disease progression.
Ethnic and racial considerations
Substantial differences in Lp(a) levels exist between ethnic groups, driven primarily by genetics. Concentrations tend to be highest in people of African descent, intermediate in South Asians, and lowest in East Asians and Europeans. When adjusted for Lp(a) concentration, cardiovascular risk appears broadly similar across ethnicities.
The journey of Lp(a) discovery
The story of lipoprotein(a) spans six decades — from an obscure immunological finding to one of the most important inherited cardiovascular risk markers in modern cardiology.
Norwegian physician Kåre Berg identifies a distinct antigen in human plasma while immunising rabbits with LDL. He names it "Lp(a)."
Kostner and colleagues purify Lp(a), noting its structural similarity to LDL but with additional protein components.
Researchers discover that apo(a) shares structural homology with plasminogen, suggesting potential roles in both atherosclerosis and thrombosis.
The LPA gene is cloned and sequenced, explaining the genetic basis of apo(a) size polymorphism and the wide variation in Lp(a) levels between individuals.
Genome-wide association studies definitively link LPA gene variants to coronary heart disease risk, confirming Lp(a) as a causal risk factor.
Lp(a) is implicated in calcific aortic valve stenosis through Mendelian randomisation, expanding its clinical significance beyond coronary artery disease.
Tsimikas et al. publish the pelacarsen phase 2 trial in the NEJM, demonstrating ≥80% Lp(a) reduction — the first proof that potent, targeted Lp(a) lowering is possible.
Phase 3 cardiovascular outcome trials for pelacarsen (Lp(a)HORIZON), olpasiran (OCEAN(a)), and lepodisiran (ACCLAIM-Lp(a)) are active. Results are expected from 2026.
Genetics and epidemiology
Lp(a) levels are primarily governed by genetic factors, with heritability estimates of 70–90%. The LPA gene on chromosome 6q26-27 is the dominant determinant. The critical feature is the kringle IV type 2 (KIV-2) copy number variant, which ranges from 3 to over 40 copies. Smaller apo(a) isoforms (fewer KIV-2 repeats) are associated with higher Lp(a) levels and greater cardiovascular risk. Additional single nucleotide polymorphisms — particularly rs10455872 and rs3798220 — account for up to 36% of variation in Lp(a) levels independently of KIV-2 copy number.
Lp(a) levels display a highly skewed distribution across populations, with a long right tail. In Caucasians, the median is approximately 12 mg/dL, with about 20% exceeding 50 mg/dL. First-degree relatives of individuals with high Lp(a) have roughly 1.8-fold increased probability of elevation. This strong genetic determination explains why Lp(a) is best thought of as an inherited lifetime risk marker rather than a lifestyle-responsive cholesterol fraction.
Structure and function
Lp(a) consists of an LDL-like lipid core — cholesteryl esters, triglycerides, phospholipids — with both apoB-100 and apolipoprotein(a) attached via a disulfide bond. Unlike LDL, which forms from VLDL metabolism in the bloodstream, Lp(a) is synthesised and secreted as a complete particle by hepatocytes. Apo(a) contains multiple kringle IV domains (with the KIV-2 domain exhibiting size polymorphism), a single kringle V domain, and a protease-like domain that is structurally similar to plasminogen but enzymatically inactive.
The size polymorphism of apo(a) results in Lp(a) particles of varying molecular weights (300–800 kDa), with an inverse correlation between apo(a) size and plasma concentration. Each Lp(a) particle carries one ApoB-100 molecule, placing Lp(a) at the intersection of inherited risk and atherogenic particle biology — one reason ApoB measurement can be informative when standard lipid measures and overall risk appear discordant.
Pathophysiology
From a practical standpoint, Lp(a) appears to increase cardiovascular risk through a combination of atherogenic particle burden, oxidised phospholipid biology, inflammation, and impaired fibrinolysis.
Atherogenic effects
Lp(a) particles penetrate the arterial intima and bind to extracellular matrix components. Their long residence time promotes cholesterol deposition and plaque growth. Macrophage uptake of oxidised Lp(a) drives foam cell formation and production of pro-inflammatory cytokines including interleukin-6 and TNF-α.
Pro-thrombotic effects
Apo(a) competes with plasminogen for fibrin binding, reducing fibrinolysis and promoting thrombus growth — particularly important at sites of plaque rupture. Lp(a) also enhances platelet aggregation in the presence of subthreshold agonists.
Calcific aortic valve disease
Oxidised phospholipids carried by Lp(a) trigger osteogenic differentiation in valvular cells, promoting calcification. Microthrombi deposition in areas of high-velocity flow, such as the aortic valve, contributes further to disease progression.
Synergistic effects
Lp(a) exerts synergistic effects with other risk factors. The combination of elevated Lp(a) and elevated LDL-C significantly heightens coronary heart disease risk. In patients with familial hypercholesterolaemia, co-elevated Lp(a) compounds cardiovascular risk substantially.
Future directions and unanswered questions
The major unanswered question is no longer whether Lp(a) is causal, but whether large pharmacologic reductions in Lp(a) will translate into fewer cardiovascular events across different risk groups. The phase 3 outcome trials currently underway will address this directly. If positive, they would validate the "Lp(a) hypothesis" and likely lead to regulatory approval of the first Lp(a)-specific therapies — potentially as early as 2026–2027 for pelacarsen.
Other important questions include: the optimal level of Lp(a) reduction needed for clinical benefit; whether Lp(a) lowering is valuable in primary prevention (current outcome trials focus primarily on secondary prevention); the role of Lp(a)-lowering in slowing aortic valve calcification; and whether there are unintended consequences of sustained, near-complete Lp(a) suppression, such as effects on wound healing or immune function.
Conclusion
Lipoprotein(a) is one of the most important inherited cardiovascular risk markers in contemporary preventive cardiology. It does not replace LDL-C, ApoB, PREVENT, or coronary calcium scoring — it complements them. In practice, the value of measuring Lp(a) lies in identifying hidden lifelong risk and using that information to individualise prevention earlier and more effectively.
Frequently asked questions
Current guidance recommends measuring Lp(a) at least once in adulthood, because it can identify inherited cardiovascular risk that is not visible on a standard lipid panel. Testing is particularly important in people with a family history of premature heart disease, familial hypercholesterolaemia, or unexplained residual risk despite well-controlled LDL-C.
A practical high-risk threshold is ≥125 nmol/L (≥50 mg/dL), although risk increases along a continuum rather than at a single cutoff. Levels around 250 nmol/L (~100 mg/dL) are associated with roughly double the long-term risk. Very high levels in the context of other risk factors warrant particularly aggressive management.
Lifestyle measures do not meaningfully lower Lp(a) itself, because levels are overwhelmingly determined by genetics. However, diet, exercise, weight management, and smoking cessation still reduce overall cardiovascular risk and remain essential when Lp(a) is elevated.
No. Lp(a) is a specific genetically determined lipoprotein particle, while LDL-C measures the cholesterol carried within LDL particles and ApoB reflects the total number of atherogenic particles. Each provides different but complementary information for risk assessment.
Usually not. Because Lp(a) is predominantly genetic and stable over time, repeat testing is generally unnecessary unless there is a specific clinical reason — such as a significant change in renal function or menopause — or when monitoring response to emerging Lp(a)-targeted therapies.
Coronary artery calcium scoring is most useful when overall risk remains uncertain after standard assessment — particularly in borderline or intermediate-risk patients where Lp(a) is elevated and treatment intensity is unclear. A zero CAC can be reassuring; a high score may prompt earlier and more aggressive treatment.
References
- Koschinsky ML, et al. Use of Lp(a) in clinical practice: a 2024 focused update from the National Lipid Association. J Clin Lipidol. 2024.
- 2026 ACC/AHA clinical practice guideline for the management of blood cholesterol and prevention of ASCVD. J Am Coll Cardiol. 2026.
- Björnson E, Borén J, et al. Relative atherogenicity of Lp(a) and LDL particles. J Am Coll Cardiol. 2024.
- Tsimikas S, et al. Lipoprotein(a) reduction in persons with cardiovascular disease. N Engl J Med. 2020;382:244–255.
- O'Donoghue ML, et al. Small interfering RNA to lower Lp(a) in cardiovascular disease (olpasiran). N Engl J Med. 2022;387:1855–1864.
- Nissen SE, et al. Lepodisiran — a small interfering RNA targeting lipoprotein(a): a phase 2 randomized clinical trial. JAMA. 2025.
- Nicholls SJ, et al. Oral muvalaplin for lowering of lipoprotein(a): a randomized clinical trial (KRAKEN). JAMA. 2025.
- Nissen SE, et al. Zerlasiran — a small interfering RNA targeting lipoprotein(a): a phase 2 randomized clinical trial (ALPACAR-360). JAMA. 2025.
- Cho L, Nicholls SJ, et al. Design and rationale of Lp(a)HORIZON trial. Am Heart J. 2025;287:1–9.
- Kronenberg F, et al. Lipoprotein(a) in atherosclerotic cardiovascular disease and aortic stenosis: EAS consensus statement. Eur Heart J. 2022;43:3925–3946.
- Willeit P, et al. Baseline and on-statin treatment Lp(a) levels for prediction of cardiovascular events: meta-analysis. Lancet. 2018;392:1311–1320.
- Burgess S, et al. Association of LPA variants and Lp(a) levels with coronary heart disease and aortic valve stenosis. JAMA Cardiol. 2018;3:619–627.
- Pare G, et al. Lipoprotein(a) levels and the risk of myocardial infarction among 7 ethnic groups. Circulation. 2019;139:1472–1482.
- Trinder M, et al. Risk of premature atherosclerotic disease associated with elevated lipoprotein(a). JAMA Cardiol. 2021;6:287–295.
- Arnett DK, et al. 2019 ACC/AHA guideline on the primary prevention of cardiovascular disease. J Am Coll Cardiol. 2019;73:e285–e350.
- Khan SS, et al. Development and validation of the PREVENT equations. Circulation. 2023;148:1982–2004.
- Reyes-Soffer G, et al. Lipoprotein(a): a genetically determined, causal, and prevalent risk factor for ASCVD. Arterioscler Thromb Vasc Biol. 2022;42:e48–e60.
- Wilson DP, et al. Use of lipoprotein(a) in clinical practice: a biomarker whose time has come. J Clin Lipidol. 2022;16:e77–e95.
- Moriarty PM, et al. Lipoprotein apheresis for lipoprotein(a) and cardiovascular disease. Eur Heart J. 2016;37:3588–3595.
- Szarek M, et al. Lipoprotein(a) lowering by alirocumab reduces the total burden of cardiovascular events. Circulation. 2024;149:192–203.